Quiz Summary
0 of 5 Questions completed
Questions:
Information
You have already completed the quiz before. Hence you can not start it again.
Quiz is loading…
You must sign in or sign up to start the quiz.
You must first complete the following:
Results
Results
Your time:
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0)
0 Essay(s) Pending (Possible Point(s): 0)
| Average score |
|
| Your score |
|
Categories
- Not categorized 0%
-

Daww you didn’t pass. Keep trying. You need to score at least 65% to pass.
-

Good! You scored above 65%. You have won a bronze medal. This earns you 1 braindollar! Go for gold?
-

Very good! You scored above 80%. You have won a silver medal. This earns you 10 braindollars! Go for gold?
-

Congrats! You scored above 90%! You have won a gold medal. This earns you 100 braindollars!
| Pos. | Name | Entered on | Points | Result |
|---|---|---|---|---|
| Table is loading | ||||
| No data available | ||||
- 1
- 2
- 3
- 4
- 5
- Current
- Review
- Answered
- Correct
- Incorrect
-
Question 1 of 5
1. Question
Between ultraviolet radiation and infrared radiation, which is lower energy and which has a longer wavelength?
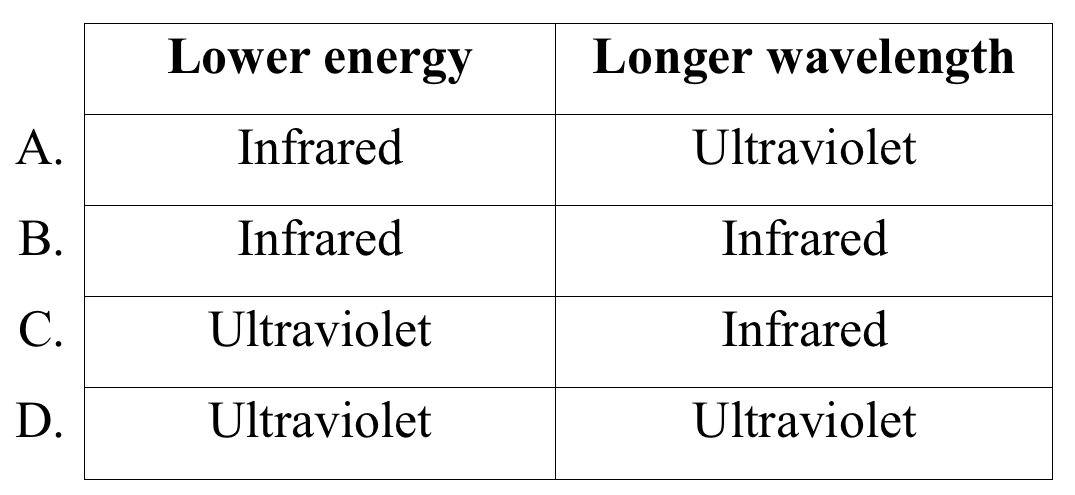 CorrectIncorrect
CorrectIncorrect -
Question 2 of 5
2. Question
An emission line spectrum is produced when:
CorrectIncorrect -
Question 3 of 5
3. Question
Which of these electron transitions would produce light in the visible region of hydrogen’s emission spectrum?
CorrectIncorrect -
Question 4 of 5
4. Question
Which of the following electron transitions in the hydrogen atom would emit the highest energy light?
CorrectIncorrect -
Question 5 of 5
5. Question
Which of these accurately explains why spectral lines in the hydrogen emission spectrum converge at higher frequencies?
CorrectIncorrect
