Quiz Summary
0 of 4 Questions completed
Questions:
Information
You have already completed the quiz before. Hence you can not start it again.
Quiz is loading…
You must sign in or sign up to start the quiz.
You must first complete the following:
Results
Results
Your time:
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0)
0 Essay(s) Pending (Possible Point(s): 0)
| Average score |
|
| Your score |
|
Categories
- Not categorized 0%
-

Daww you didn’t pass. Keep trying. You need to score at least 65% to pass.
-

Good! You scored above 65%. You have won a bronze medal. This earns you 1 braindollar! Go for gold?
-

Very good! You scored above 80%. You have won a silver medal. This earns you 10 braindollars! Go for gold?
-

Congrats! You scored above 90%! You have won a gold medal. This earns you 100 braindollars!
| Pos. | Name | Entered on | Points | Result |
|---|---|---|---|---|
| Table is loading | ||||
| No data available | ||||
- 1
- 2
- 3
- 4
- Current
- Review
- Answered
- Correct
- Incorrect
-
Question 1 of 4
1. Question
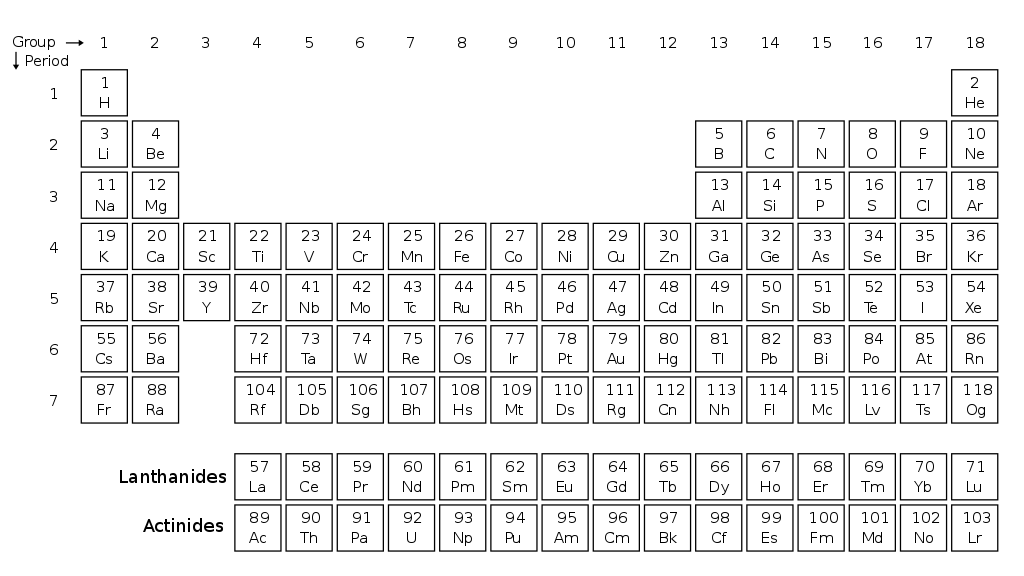
The graph shows the first five ionisation energies (IEs) of an element.
Which of these is the element?
CorrectIncorrect -
Question 2 of 4
2. Question
The table shows the first eight ionisation energies (IEs) for an element. Deduce the group of the element.
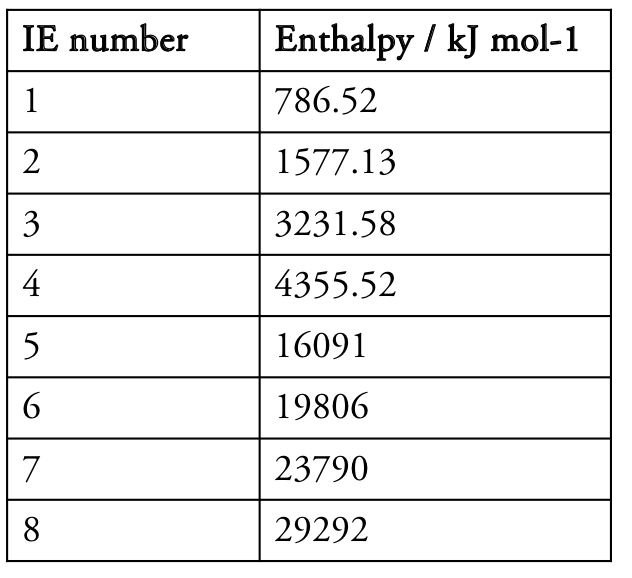 CorrectIncorrect
CorrectIncorrect -
Question 3 of 4
3. Question
Which of these is a correct explanation for the decrease in first ionization energy (IE) noted between nitrogen and oxygen?
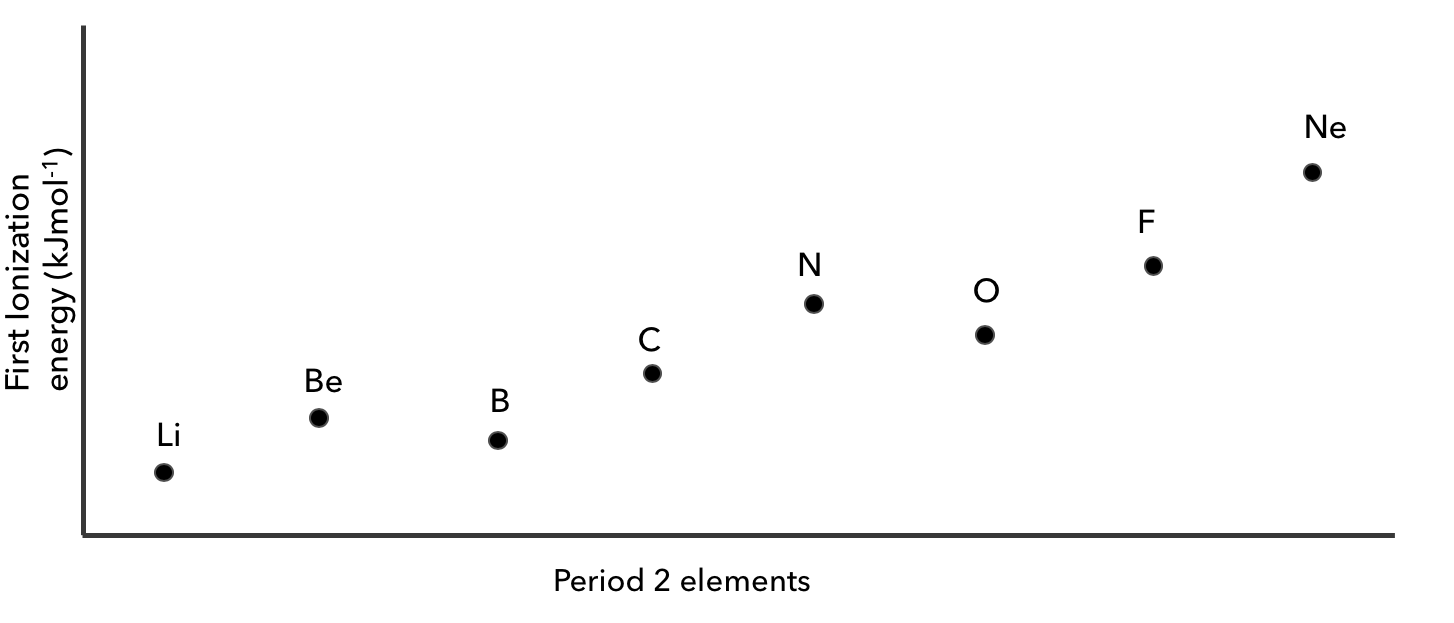 CorrectIncorrect
CorrectIncorrect -
Question 4 of 4
4. Question
Which of these is a correct explanation for the decrease in first ionization energy (IE) noted between beryllium and boron?
 CorrectIncorrect
CorrectIncorrect
